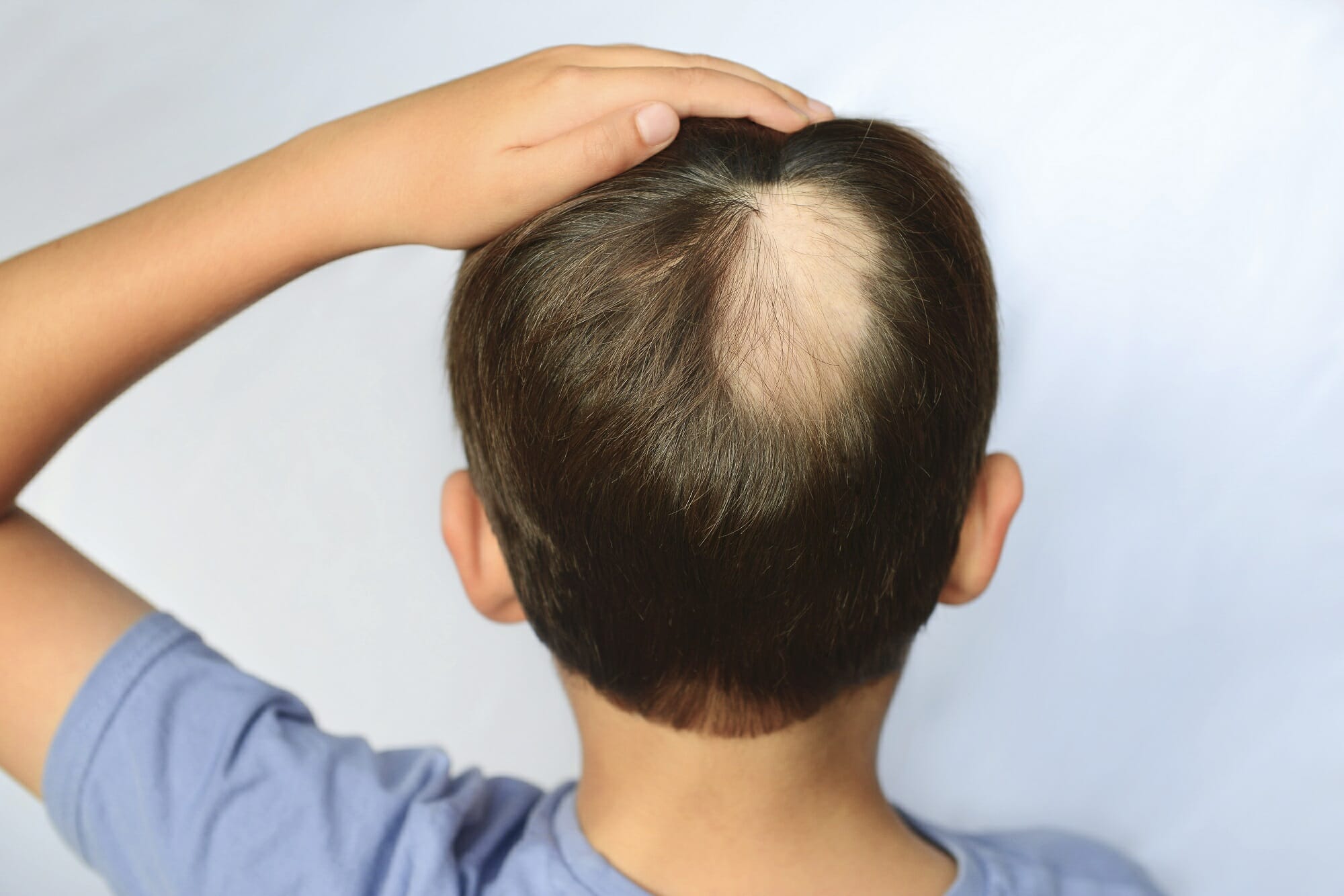
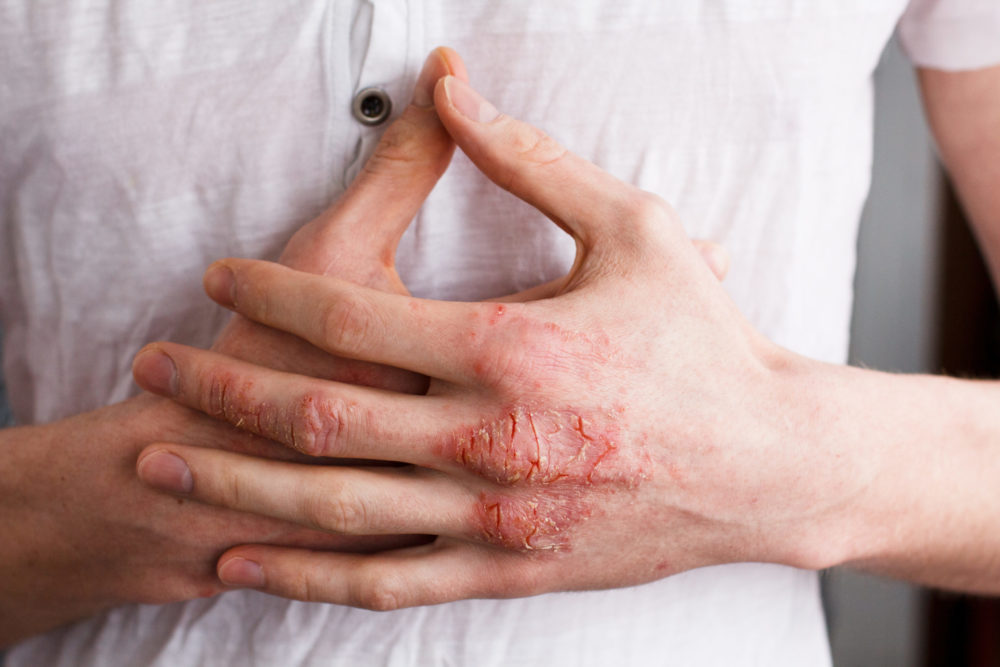
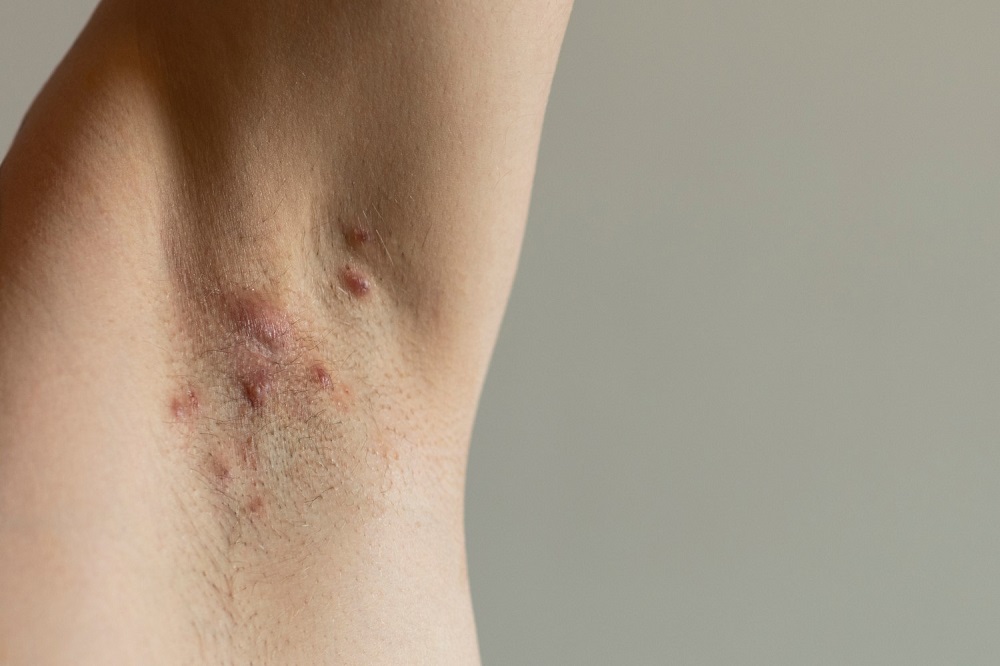
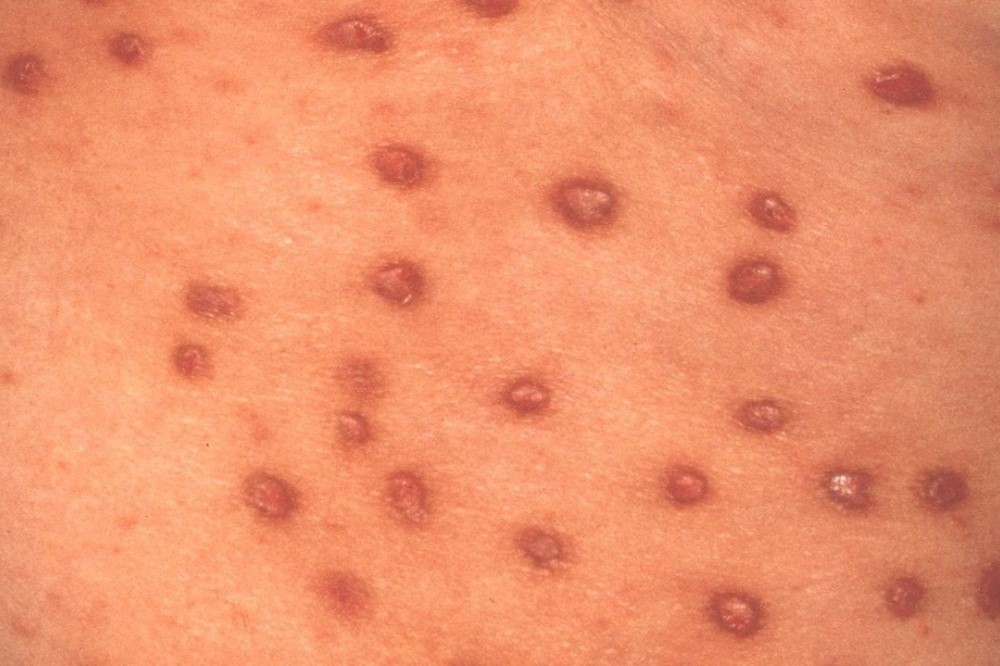
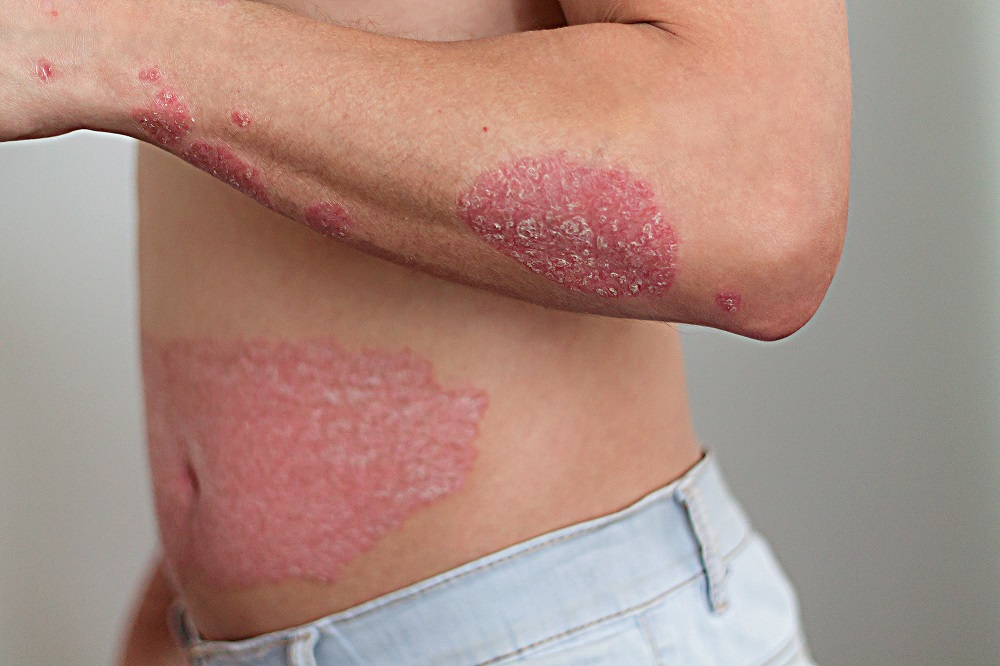
Active
Clinical Trials
Join us in creating a brighter future for generations to come.
Active Clinical Trials
Search a clinical trial by treatment type
Clinical Trials Saving Families & Lives
By participating in clinical studies or trials, you can increase the pace of research and directly improve the lives of millions dealing with life-changing diseases.

Frequently Asked Questions
A clinical trial is a research study in which volunteers participate to help develop new therapies and diagnostic procedures. Researchers use different types of trials to answer specific questions before they ask the FDA (Food and Drug Administration) to approve a medication.
For example:
- Does the medication or treatment help the condition or disease?
- What are the side effects?
- What is the best dose?
- Can an existing treatment be effective for a different condition or disease than the original one it was tested for?
- Can an existing treatment be effective in a different formulation, like a pill instead of an injection, or a cream instead of a pill?
In the U.S., clinical trials are regulated by the FDA. Researchers follow strict rules to ensure the protection of a volunteer’s rights and safety. For more information about clinical trials and treatment development, visit clinicaltrials.gov or the FDA website for more information.
Clinical trials are conducted according to a plan which is often referred to as a “protocol.” Each protocol has its own eligibility criteria on who can (or cannot) participate. Your eligibility may be based on a variety of factors such as age, current medications you take, previous treatment history, and other medical conditions.
Defining this eligibility is not only beneficial to the research study by increasing the likelihood to produce reliable data, but also benefits its volunteers by reducing the likelihood of harm. Because of this, not everyone is chosen to participate. Those who do participate in our clinical trials do so freely and of their own will.
The primary focus of a clinical trial is to build knowledge by collecting data that can help doctors and researchers better understand, diagnose, and treat diseases. If you participate in a clinical trial, you will be contributing to the cumulative effort to fight these diseases. While it depends on the specific trial you participate in, there may be a chance you do not receive the treatment under study, in the form of a placebo or an existing treatment already approved. It is important to consider your options carefully in consultation with your healthcare provider. If you do choose to participate, you will have access to expert medical care but be prepared to adhere to a schedule of visits outlined in the study protocol. Please note, it is not guaranteed that you will see an improvement in your health, and there is a possibility your disease state may progress during the study.
If you participate in a study, you will be asked to attend study visits according to the schedule in the protocol. You will need to follow instructions from the study staff, for example, on how to fill out forms or use the study medication. We will also ask you to keep us updated on any changes in your health or medications while you are enrolled in the study.”
What happens at a visit varies from study to study. Common study activities include answering questionnaires about your symptoms or how you feel, skin assessments by the provider, and taking vital signs (like blood pressure). Some studies require ECGs or blood collection. Study staff will go over all the procedures involved in a study before you decide to participate.
Your participation is entirely voluntary and there is no obligation to enroll. The research doctor will explain the potential benefits and risks to you so you can make an informed decision about participating.
Benefits
- Participating in a clinical trial may give you the opportunity to receive a new treatment early in its development and before it is available at local pharmacies.
- The research team will watch you and your health closely, which some people find reassuring
- You may help researchers learn more about skin diseases and help people in the future
Risks
- As with most medical treatments, there may be side-effects associated with the experimental treatment.
- Depending on the study design, your participation may require more time and attention than standard clinical care.
- The new treatment may not work for you